Urges Stockholders to Vote the WHITE Proxy Card “FOR ALL” of Adverum’s Three Highly Qualified, Diverse and Independent Directors:
Annual Meeting to Be Held on
The Adverum Board of Directors unanimously recommends that stockholders vote the WHITE proxy card FOR Adverum’s three highly qualified directors standing for election -
In conjunction with the definitive proxy filing, Adverum is mailing a letter to stockholders detailing the significant clinical advancements the Board and management team are executing to accelerate toward commercialization and create value for stockholders. The full text follows and can be found on the investor section of the Company's website at https://investors.adverum.com/shareholder-services/annual-meeting.
VOTE THE WHITE PROXY CARD TODAY “FOR ALL” OF ADVERUM’S THREE HIGHLY QUALIFIED, DIVERSE AND INDEPENDENT DIRECTORS
REJECT SONIC’S SELF-SERVING ATTEMPT TO TAKE CONTROL OF THE ADVERUM BOARD
Dear Fellow Stockholder,
You have an important decision to make regarding the future of your investment in Adverum. At our Annual Meeting of Stockholders to be held on
Adverum is making tremendous progress implementing plans across the business to support a successful development strategy and commercial launch. We urge you to ensure this momentum continues by supporting Adverum’s three highly qualified directors standing for election at this year’s Annual Meeting and vote your shares FOR ALL of Adverum’s director nominees -
Your vote is especially important this year given that
Over the last several years, we have maintained an open and constructive dialogue with Sonic, including working collaboratively with Sonic in May of 2019 to appoint to our Board two new directors proposed by Sonic. The Board and management team of Adverum spent significant time, both before and after the settlement agreement in 2019, engaging with Sonic’s principal,
Despite Adverum’s good faith efforts to take feedback and incorporate Mr. Kam’s perspectives as reasonably as possible for over the past two years, it has now become clear that
Adverum’s significant clinical advancements with ADVM-022 have been guided by a strong Board and leadership team that has been meaningfully refreshed over the last two years. Given the positive action and progress underway, Sonic’s proxy contest is unnecessary, distracting, costly and not in the best interest of all stockholders. Sonic’s proposed nominees, if elected, together with its two designees appointed in 2019, would constitute more than half of the Board. We do not believe allowing an approximately 7% stockholder to hand-pick a majority of the Board is in the best interest of our stockholders, our patients or any of our other stakeholders.
While Sonic has openly criticized Adverum’s leadership and strategy, Sonic has not articulated a single strategic plan or provided details on what it intends to do with your company if Sonic gains control of the Board. All
Your vote on the WHITE proxy card in advance of our Annual Meeting of Stockholders is critically important, no matter how many shares you own. Not doing so would put at risk our accelerating momentum and execution of a clear strategic plan to establish our leading ocular gene therapy program as a one-time treatment that preserves patient sight for life.
Transformative Gene Therapy
Adverum has an incredible opportunity ahead of it, and one that we work tirelessly every day to bring to fruition for the benefit of patients. Our market opportunity is large and rapidly growing:
- Approximately nine million patients live with wet age-related macular degeneration (AMD) or diabetic macular edema (DME) treatable with anti-VEGF worldwide1; and
$11.5 billion was spent globally on approved anti-VEGF therapies in 2020,2 with 12% of theU.S. Medicare Part D budget spent on anti-VEGF therapies annually3.
The current standard of care for wet AMD and DME requires an eye injection every four to eight weeks, often requiring a full day for treatment and recovery, in order to avoid vision loss. Therefore, it should come as no surprise that Adverum’s extensive market research and frequent discussion with retina specialists and patients reveal that the largest unmet need for patients with wet AMD is for a durable treatment that reduces the frequency of injections and provides increased efficacy, improved ease of administration and better control of macula fluid.
ADVM-022, our program designed to treat wet AMD and DME with a single in-office intravitreal injection, is the only investigational non-surgical treatment option that has demonstrated long-term durability up to two years. We believe that ADVM-022 is uniquely positioned, unlike any other treatment on the market or in development, as an advanced gene therapy product with the potential to be a one-and-done approach for millions of people living with wet AMD and DME globally.
Based on the results in our OPTIC trial to date and our constructive discussion with the
To date, ADVM-022 in wet AMD has demonstrated favorable efficacy and safety profiles. In the OPTIC trial, patients who received 2 x 10^11 vg/eye of ADVM-022 experienced an 85% reduction in annualized anti-VEGF injections and two-thirds remained supplemental anti-VEGF injection free with median follow up of 48 weeks4.
The safety of patients is our top priority. While data shows that ADVM-022 continues to be well tolerated with ocular cell grades and steroid eye drop use decreasing over time, we have convened multiple advisory boards of experts to optimize the prophylaxis regimen and are following our advisors’ recommendations. We are committed to pursuing multiple paths to improve results with respect to inflammation given the treatment durability observed beyond two years in patients who received a single injection of ADVM-022.
In addition, Adverum announced in
Accelerating Toward Commercialization
Your Board and management team are taking decisive action to accelerate our ADVM-022 development and commercialization plans to launch our first mass-market gene therapy to treat millions of patients with wet AMD and DME if ADVM-022 receives FDA approval. We are focused on execution, with the goal of delivering this transformative gene therapy to patients globally as early as possible.
In
In addition, Adverum has all process development, assay development and quality control in-house and is partnering with contract manufacturing organizations to manufacture clinical and additional commercial supply. We use a highly scalable baculo/Sf9 suspension process, having scaled up from 50 liters to 200 liters, and are now in the process of scaling up to 1,000 liters.
Importantly, given the complexity of gene therapy manufacturing, controlling the entirety of the manufacturing process has proven to be critical in maximizing the value of the therapy long-term. Owning an in-house GMP facility that can provide dedicated commercial supply at scale is a core differentiator for gene therapy companies, as it meaningfully de-risks the ability to get these critical therapies to patients. Gene therapy companies with owned manufacturing facilities are further rewarded for this de-risking through higher market valuations and greater strategic interest over the long-term.
Adverum maintains a disciplined approach to capital allocation and will continue to invest in advancing ADVM-022 toward commercialization and further building out our manufacturing capabilities – with the goal of creating long-term value for our stockholders.
Delivering Value for Our Stockholders
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/2496e46f-9eed-46c0-8dc8-5a24a3114549
Even before commercialization, Adverum’s efforts have already delivered value for our stockholders. Adverum has delivered five- and three-year total stockholder return of 91% and 70% respectively, significantly outperforming gene therapy peers.5
As Adverum continues to report positive interim data and continues to achieve pre-commercialization, clinical and development milestones throughout the rest of 2021 and beyond, we expect to drive significant value for our stockholders.
Importantly, additional clinical data will be presented for Cohorts 1-4 in the OPTIC clinical trial of ADVM-022 intravitreal injection gene therapy in wet AMD during the
Your Recently Refreshed Board is Highly Qualified, Engaged and Diverse: Seven New Independent Directors Added in the Last Two Years, Including Two of Sonic’s Designees
Adverum has an experienced, well-rounded and independent Board that benefits from the right mix of skills to position it for success as we navigate upcoming clinical and operational milestones. Our world-class directors are industry leaders that are actively engaged in overseeing Adverum’s strategic execution and are holding management accountable.
Seven of our nine continuing directors are independent, eight are non-employees and all of our directors bring expertise critical to our business, including gene therapy and other drug development, ophthalmology, global commercialization, R&D, policy and managed care expertise, as well as finance and accounting, governance and risk management.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/d599c5d4-e40d-41c0-a843-7ebe9b27037d
The Adverum Board is also committed to ongoing refreshment to ensure a best-in-class and diverse make up. In the last two years, the Board has appointed seven new non-employee directors, three in
While Sonic’s nominees are not right for Adverum, the Board is committed to continuing its refreshment efforts and has made it a priority to recruit a high-quality director with product and commercial gene therapy experience to be named in 2021. Notably, we have engaged with our stockholders about top candidates and already interviewed candidates to fill this slot, and strongly believe that this is a superior process to relying solely on input from one stockholder.
Your Board’s nominees for the 2021 Annual Meeting –
Ms. Svoronos is a commercialization expert and has three decades of global biopharmaceutical industry experience, spanningthe United States ,Canada ,Europe , andAsia . During her over two-decade career at Merck,Ms. Svoronos led and/or was involved in dozens of pharmaceutical product launches in key markets around the world, many of which were mass market opportunities.Ms. Svoronos serves on the Board of four companies, including Global Blood Therapeutics, Inc., and PTC Therapeutics.Dr. Tuckson is an esteemed healthcare executive and brings extensive policy knowledge from his experience in multiple facets of the healthcare industry, including a 13-year tenure in senior leadership positions at UnitedHealth Group, where he served as chief of medical affairs. He has a strong understanding of the complex reimbursement environment and proven track record of enhancing patient access, as exemplified by his recent COVID-19 vaccine efforts in disproportionally impacted minority populations.Dr. Woiwode , an Adverum director since 2016, has been withVersant Ventures since 2002, serving as a venture partner since 2008 and a managing director since 2014.Versant Ventures is a significant stockholder of Adverum.Dr. Woiwode has served in a number of operating roles, most recently as the chief operating officer ofOkairos , which was sold to GlaxoSmithKline plc.Dr. Woiwode serves as a director at Aligos Therapeutics, Inc., Gritstone Oncology, Inc., Passage BIO, Inc. and several private life sciences companies.
Our commitment to value creation, diversity and ESG principles is embedded in our core and helps drive long-term value creation for stockholders. The entire Adverum Board is dedicated to ensuring the management team and long-term strategy is mission driven. Public health is the essence of who we are, and the Board considers this one of our most important ESG values.
Further, our ability to help people extends well beyond working to treat vision loss and eye disorders. Our people are our brain trust. We believe our culture is a key advantage and we are committed to supporting our employees. We believe brands are built from the inside out, and our employees are the cornerstone of our business.
Refreshed, World-Class Management Team in Place to Navigate Upcoming Milestones and Successfully Drive Toward Commercialization
We have a refreshed management team in place to drive our strategy, including Chief Executive Officer
In addition, a new Chief Scientific Officer has accepted Adverum’s offer and will be announced in the coming weeks, and we are conducting a comprehensive search process to identify our next Chief Medical Officer.
Adverum Has a Long History of Good
Adverum has carried on an active dialogue with
During this long engagement,
Mr. Kam expressed his desire forMr. Chalberg to be the CEO of Adverum, stating to one of the directors he placed on our Board in 2019 that “He [Dr. Chalberg ] really should be Executive Chairman, CEO, or whatever but has other commitments and knows that his association with Avalanche 1.0 does not help shake the Avalanche 2.0 image.’’ Two of Mr. Kam’s nominees also have ties to Avalanche, one as an employee and one as a scientific advisory board member.Mr. Kam repeatedly proposed that Adverum divert resources to pursue a$20 million lead investment in the initial financing round of a start-up gene therapy company using a different technology in a different therapeutic area from what Adverum is pursuing. Notably, to our knowledge, this company has not been financed or progressed meaningfully.
Despite Mr. Kam’s questionable actions, your Board and management team have remained committed to open engagement and to considering Mr. Kam’s ideas. In fact, on
Sonic’s Attempt to Replace Three Highly Qualified and Diverse Independent Directors with Its Own Nominees Puts at Risk Operational Execution and Is Not in the Best Interests of Stockholders
Not only would Sonic’s nominees, if elected, together with its two nominees appointed in 2019, constitute more than half of the Board, their election would significantly diminish the diversity of expertise and experience on the Board that has been so critical to our recent efforts towards commercial readiness. We believe that allowing Sonic to hand-pick a majority of the Board would significantly disrupt our progress toward achieving our upcoming milestones and delay the value opportunity of ADVM-022.
Sonic is putting at risk Adverum’s laser focus on operational execution as well as our ability to retain and recruit world-class talent. While Sonic is focused seemingly on reassembling a team from the past, Adverum is staying focused on the future and accelerating toward commercialization. Stockholders should not allow Sonic to impede our momentum.
Your Vote Is Extremely Important! Vote the WHITE Proxy Card Today to
Vote the Enclosed WHITE Proxy Card Today “FOR ALL” Three of Adverum’s Highly Qualified and Diverse Director Nominees
Adverum is well-positioned to capitalize on the opportunities ahead. Your Board and management team are focused on accelerating the development and future commercial launch plans for ADVM-022, and we firmly believe we have the right directors in place to do just that.
We urge you to use the enclosed WHITE proxy card to vote today "FOR" ALL three of Adverum's nominees listed on the WHITE proxy card:
We appreciate your continued support as we work to drive value for ALL stockholders.
Sincerely,
The Adverum Board of Directors
Advisors
About
Forward-looking Statements
Statements contained in this press release regarding the events or results that may occur in the future are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Such statements include, but are not limited to, statements regarding Adverum’s expectations that its two global Phase 3 trials in wet AMD will initiate in parallel in the fourth quarter of 2021, that it will submit a Biologics License Application in 2024, that it expects to present clinical data from the INFINITY trial in the second half of 2021, that its commercial facility in
Important Information
1 MarketScope Retina Report for 2020 in US, EU, JP
2 Based on 2020 public filings from Regeneron, Roche and Novartis
3 Medicare Spending on Anti-Vascular Endothelial Growth Factor Medications,
4 Adverum press release on
5 FactSet as of
Investor Relations ContactMyesha Lacy Vice President, Investor Relations andCorporate Communications Adverum Biotechnologies, Inc. T: 650-649-1257 E: mlacy@adverum.com OrInnisfree M&A Incorporated Scott Winter /Gabrielle Wolf T: 212-750-5833 Media ContactAndrea Cohen Sam Brown Inc. T: 917-209-7163 E: andreacohen@sambrown.com OrJoele Frank ,Wilkinson Brimmer Katcher Dan Moore T: 212-355-4449
![]()
Adverum 1, 3 and 5-Year Total Stockholder Returns
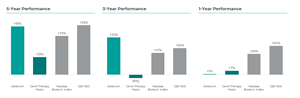
Adverum 1, 3 and 5-Year Total Stockholder Returns
Adverum Director Skills & Experience Matrix
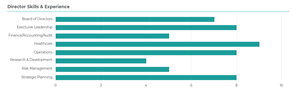
Adverum Director Skills & Experience Matrix
2021 GlobeNewswire, Inc., source


